DSC Analysis Explained: Basics for Pharma R&D
Introduction
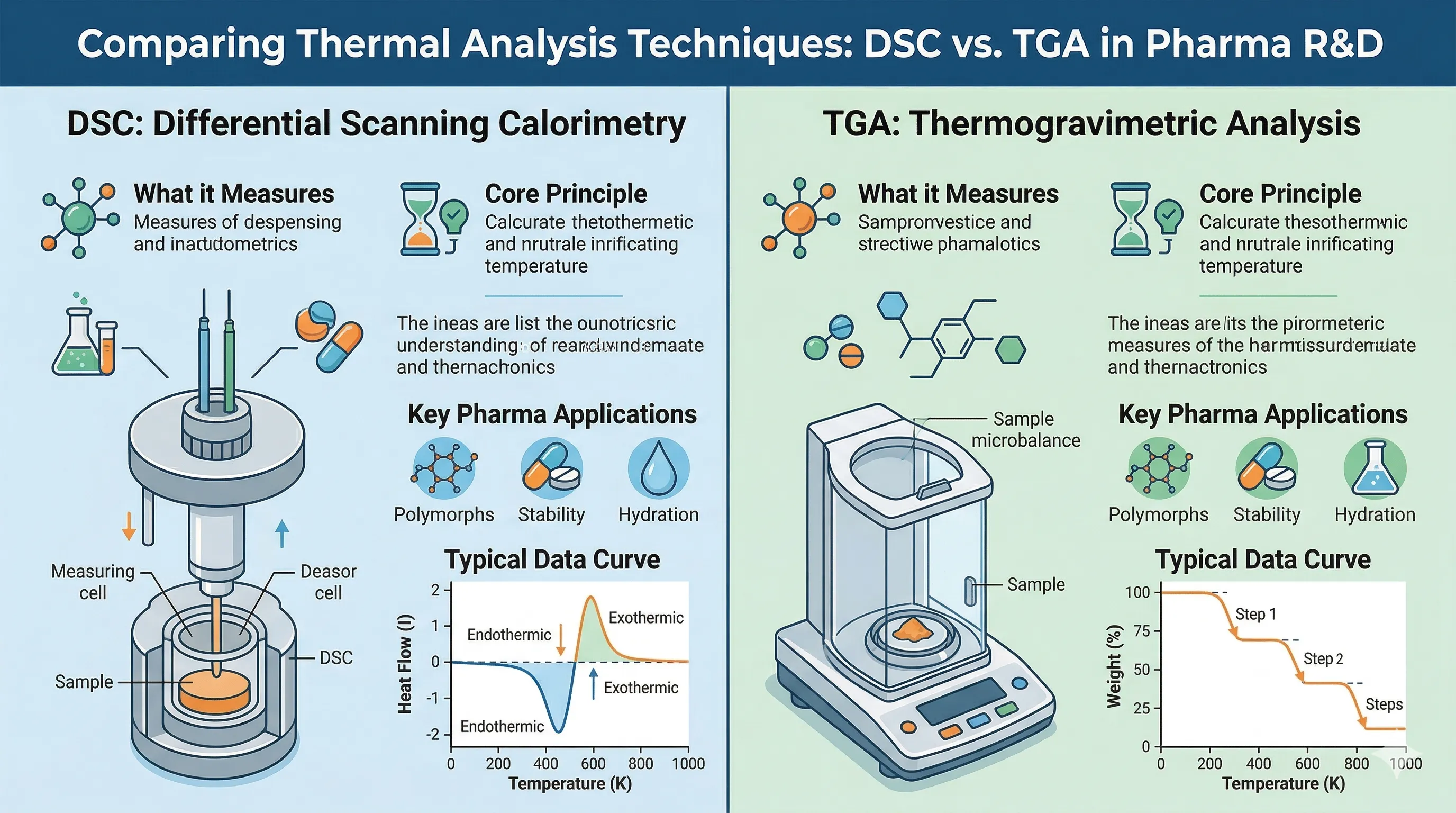
In the fast-paced world of pharmaceutical research and development, understanding the thermal behavior of drug substances and formulations is non-negotiable. Differential Scanning Calorimetry (DSC) stands as the gold standard technique for this purpose. DSC provides a fingerprint of the thermodynamic properties of materials, unlocking crucial data concerning melting points, glass transitions, crystallization events, and polymorphic characteristics.
For pharmaceutical scientists, this data is the cornerstone of drug formulation, stability profiling, and manufacturing quality control. A minor phase change or polymorphic instability can compromise drug efficacy and patient safety. By characterizing these thermal variations rapidly and accurately, DSC bridges the gap between raw molecular synthesis and approved, market-ready therapeutics. This guide will walk you through the fundamentals, key applications, relevant case studies, and industry best practices for DSC in modern labs.
The Theory Behind DSC
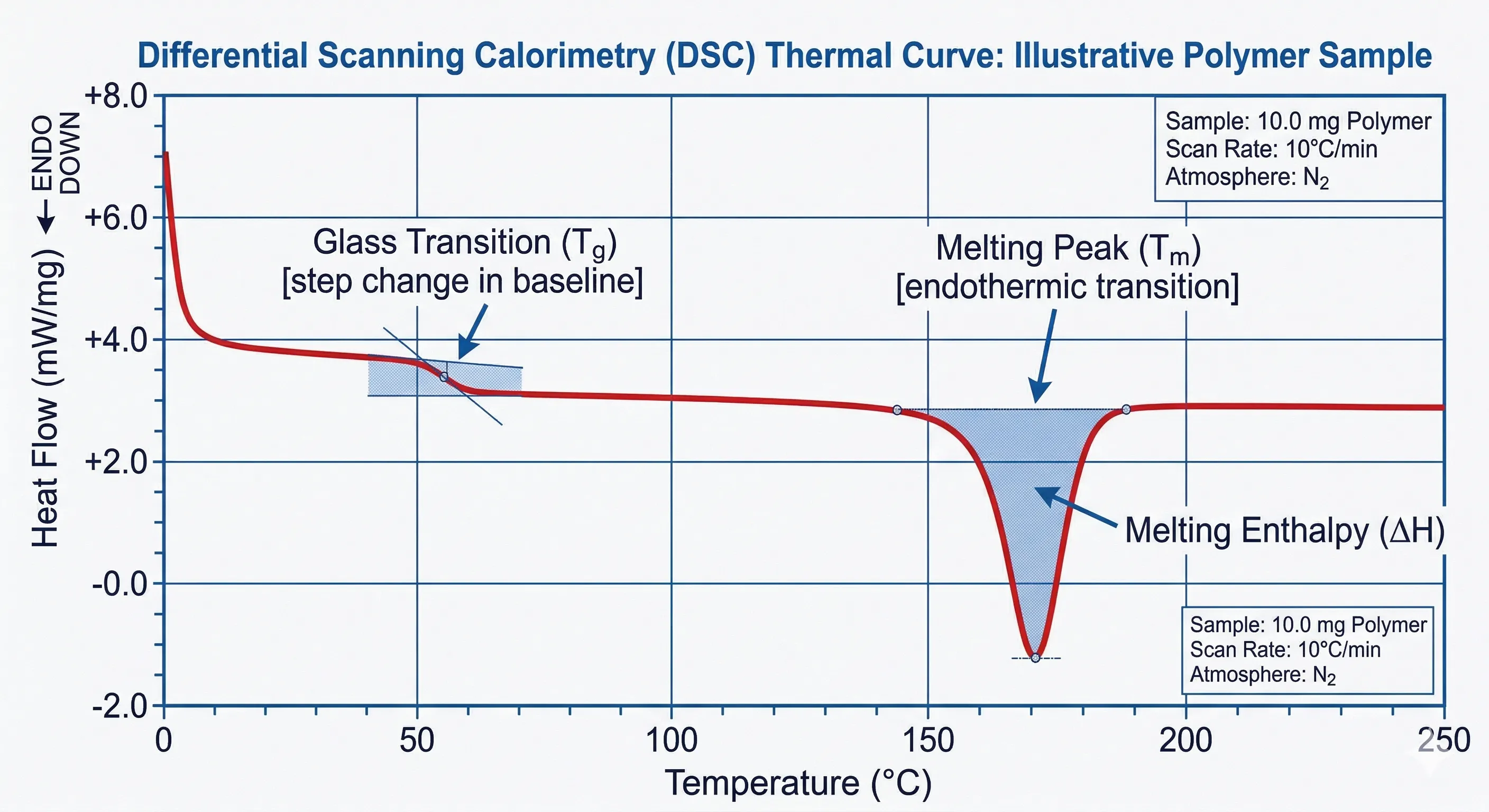
Differential Scanning Calorimetry works on a beautifully elegant principle: measuring the difference in the amount of heat required to increase the temperature of a sample and a reference as a function of temperature. Both the sample and reference are maintained at nearly the same temperature throughout a controlled heating program.
When the sample undergoes a physical transformation, such as phase transitions (melting or crystallization), more or less heat will need to flow to it than the reference to maintain both at the same temperature. By tracking these heat flow variations, a thermal curve (or thermogram) is produced. Endothermic events, where the sample absorbs heat (like melting), appear as peaks indicating energy absorption. Exothermic events, such as crystallization, appear in the opposite direction.
Key Applications in Pharma R&D
DSC is ubiquitous in pharmaceutical laboratories due to its immense versatility:
1. Polymorphism Studies: Many Active Pharmaceutical Ingredients (APIs) can exist in multiple crystalline forms (polymorphs), each with different solubility and bioavailability profiles. DSC accurately identifies these polymorphs.
2. Glass Transition Temperature (Tg) Determination: Critical for amorphous solid dispersions. The Tg dictates the storage conditions and stability of an amorphous drug formulation.
3. Purity Determination: Based on the melting point depression principle, DSC can estimate the absolute purity of a crystalline substance rapidly, often acting as a complementary technique to HPLC.
4. Drug-Excipient Compatibility: Pre-formulation studies use DSC to detect thermal interactions between an API and potential excipients, ensuring no detrimental reactions occur during manufacturing or storage.
Case Study: Optimizing Formulation Stability

Consider a recent project involving a poorly soluble API formatted into an amorphous solid dispersion (ASD) to enhance bioavailability. Early stability testing indicated physical instability, with the formulation gradually recrystallizing.
By employing high-sensitivity DSC from METTLER TOLEDO, the research team identified that the formulation's initial Glass Transition Temperature (Tg) was too close to standard storage conditions. Over time, ambient moisture plasticized the polymer matrix, further suppressing the Tg. The DSC thermograms clearly showed the onset of crystallization long before it became visible via X-Ray Diffraction (XRD). Armed with this thermal insight from the DSC, the formulation was adjusted to include a higher Tg polymer, actively resolving the stability crisis before clinical trials.
Best Practices for Accurate DSC Testing
To ensure the integrity and reliability of DSC data, adhering to strict best practices is essential:
- Sample Preparation: Ensure excellent thermal contact by pressing the sample flat against the bottom of the pan. Use the optimal sample size—typically 2-5 mg for pharmaceuticals.
- Atmosphere Control: Always use a controlled purge gas, typically dry nitrogen, to prevent sample oxidation and moisture interference during the run.
- Regular Calibration: Regularly calibrate the instrument for both temperature and heat flow using standard reference materials (like Indium and Zinc).
- Adherence to Pharmacopeia: Follow guidelines established by recognized regulatory bodies, such as USP <891>.
Related Resources
To dive deeper into the technical specifications and guidelines regarding DSC, explore these authoritative resources:
Conclusion
Differential Scanning Calorimetry is more than just a regulatory reporting requirement; it is a fundamental tool for pharmaceutical innovation. From discovering robust polymorphic forms to ensuring the long-term stability of complex formulations, DSC equips scientists with the thermal clairvoyance needed to deliver safe, effective therapeutics to market. As equipment technology advances, such as the precision found in modern METTLER TOLEDO systems, the depth of insight provided by DSC will only continue to scale.
Need Expert Thermal Analysis?
Our METTLER TOLEDO-equipped lab delivers precise, NABL-traceable results for DSC, TGA, RC1, and more.
Book Free ConsultationShare Article
Related Articles
Glass Transition Temperature (Tg): Why it Matters in Pharma
Understand why the Glass Transition Temperature (Tg) is the most critical parameter for ensuring the stability of amorphous solid dispersions in pharmaceuticals.
PharmaceuticalsEvaluating the Shelf Life of Lyophilized Pharmaceuticals
Explore how DSC and TGA thermal analysis techniques prevent collapse, detect moisture ingress, and guarantee the shelf life of lyophilized biologic drugs.
Thermal AnalysisDifferential Thermal Analysis (DTA): History and Modern Use
Explore the legacy and highly specialized modern applications of Differential Thermal Analysis (DTA), the high-temperature predecessor to standard DSC.